Suggested Articles
Advanced Simulation May Change Product Manufacturing for the Better
Featured
Hot Topics
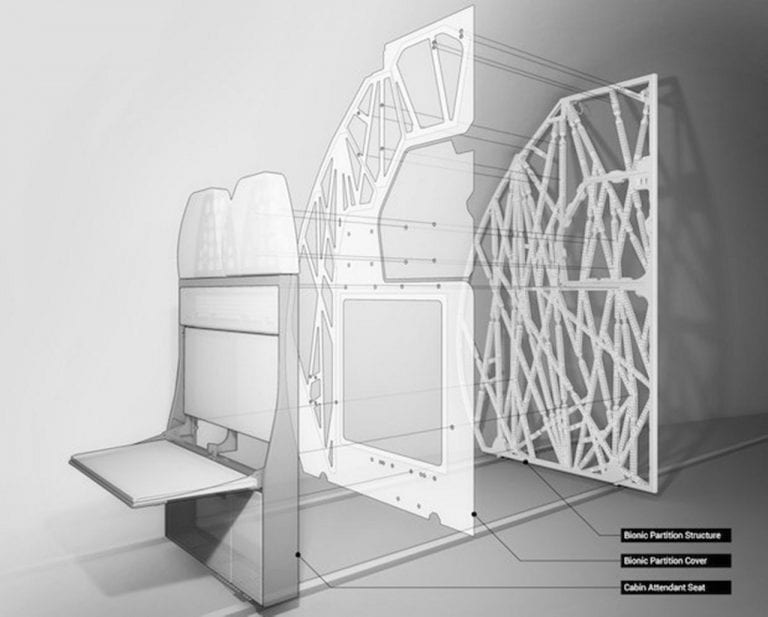
There is a class of things that merge form and performance into labyrinthine lines that no person could have created. They are designed by artificial intelligence nudged toward...
