Suggested Articles
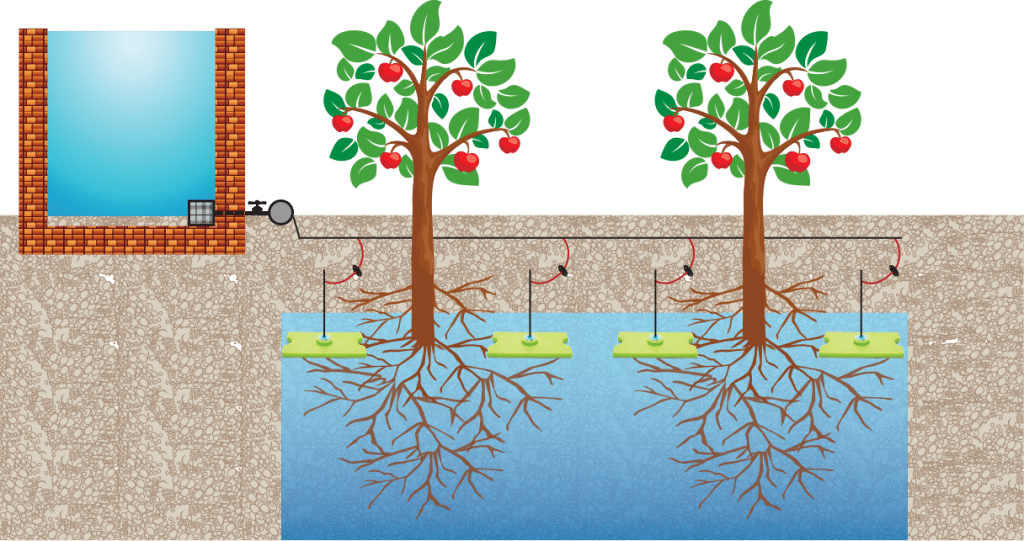
Ten Technologies for Farming in Dry Climates
Featured
Hot Topics
Farmers in dry climates manage to coax crops from sandy soil and water them with rain and even fog. We mined our Solutions Library and network of experts for...
No Comments.