Suggested Articles
Lean Manufacturing of Disease Screening Strips: How a Stamp Simplifies...
Featured
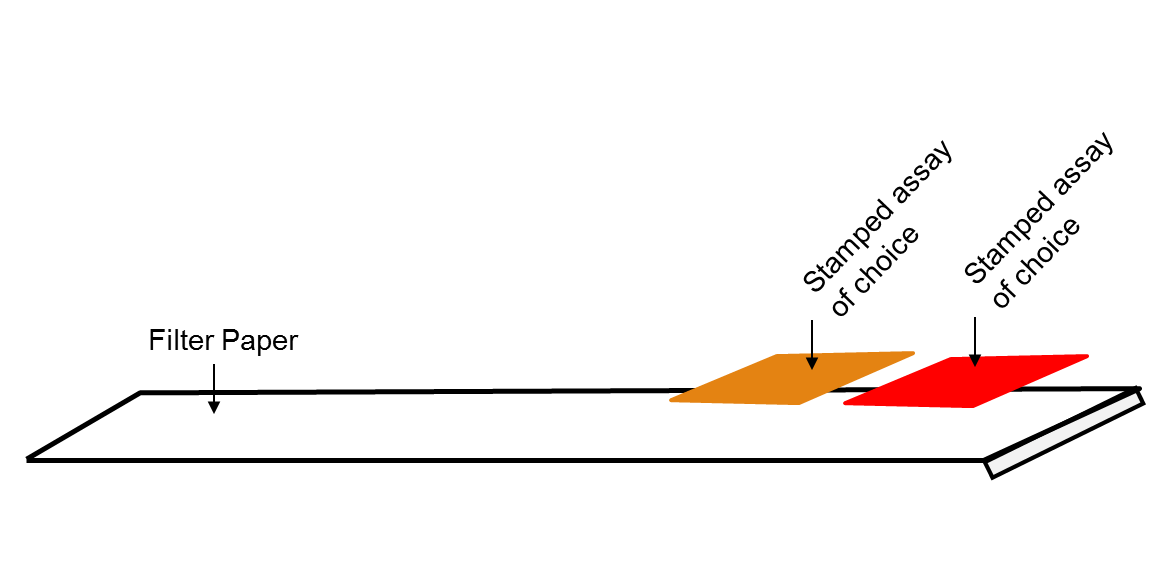
Using paper, a couple of assays and a simple foam stamp, a new manufacturing technique can make disease screening strips more affordable. Treatable diseases often go undetected in developing countries due...
