
Agriculture
January 16, 2024
Integrated Wetland Technology
Read SolutionImplemented by
Emergy Enviro Pvt. Ltd
Updated on November 22, 2023
·Created on October 3, 2020
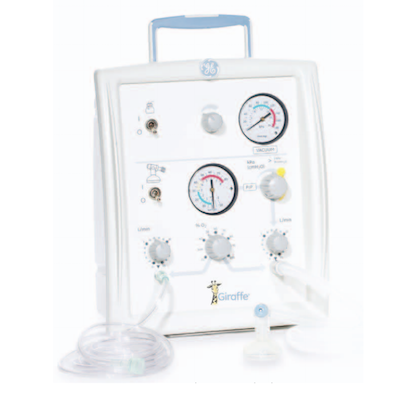
Quick neonatal resuscitation delivery.
The Integrated Resuscitation Systems device is a quick, all-in-one system to quickly provide neonatal resuscitation. The device provides effective airway management, gentle ventilation, and pulse oximetry. The device consists of a bag and mask, and a T-piece design.
Target Regions
Europe
Target SDGs
SDG 3: Good Health and Well-Being
Market Suggested Retail Price
$4,070.00
Target Users (Target Impact Group)
Community, Small and Medium-sized Enterprises
Distributors / Implementing Organizations
General Electric Healthcare Systems
Regions
Europe
Manufacturing/Building Method
Unknown
Intellectural Property Type
Patent Protected
User Provision Model
Interested users should directly contact the manufacturer for details.
Distributions to Date Status
Over 25,000 devices have been distributed to date.
Design Specifications
The product can take a hospital pipeline or a cylinder as gas input. The product features a gas blender that allows the output oxygen concentration to vary between 21-100% O2. The device can largely be operated with one hand. The airway management features a fully integrated suction and the positive pressure delivery can be monitored during delivery. The system can be mounted on a roll stand and a GCX.
Product dimensions are: 13.4 x 9.7 x 9.8 in. The product weighs 19.8 lbs with Blender.
Technical Support
Provided by the manufacturer.
Replacement Components
The T-piece is a single use item and needs replacement between patients, which can be purchased from the designer. Cylinders for oxygen supply require replacement when their levels are low.
Lifecycle
5+ years
Manufacturer Specified Performance Parameters
Manufacturer specified performance targets include: quick, integrated features, one-handed ergonomic operation, and easy to use.
Vetted Performance Status
The system has undergone integration, performance, safety, usability, and biocompatibility testing according to U.S FDA guidelines.
Safety
A PIP safety valve is used is used in the system for easy one hand activation by pressing the PIP knob release.
Complementary Technical Systems
Compatible with Panda and Giraffe Warmers. GE (the designer) offers system holding kits such as a GCX arm and roll stand, as well as a T piece disposable circuit for purchase.
Academic Research and References
M. E. Cavicchiolo et al., 2018, Decision making and situational awareness in neonatal resuscitation in low resource settings, Resuscitation, vol. 134, pp. 41–48.
S. K. Berkelhamer, B. D. Kamath-Rayne, and S. Niermeyer, 2016, Neonatal Resuscitation in Low-Resource Settings, Clinics in Perinatology, vol. 43, no. 3, pp. 573–591.
S. N. Wall et al., 2009, Neonatal resuscitation in low-resource settings: What, who, and how to overcome challenges to scale up?, in International Journal of Gynecology and Obstetrics, vol. 107, pp. S47–S64.
Compliance with regulations
The system was designed according to the American Academy of Pediatrics' (AAP) latest Neonatal Resuscitation Program Guidelines. The product is in compliance with the U.S. FDA.
Other Information
The Giraffe and Panda i-Res Infant Warmers, complementary systems to the Integrated Resuscitation Systems, underwent a Class I FDA Recall in 2019 for certain products issued between 2008-2019 due to complaints related to bedside panels/latches.

Agriculture
January 16, 2024
Implemented by
Emergy Enviro Pvt. Ltd

Agriculture
February 5, 2024
Implemented by
Gradian Health Systems

Agriculture
March 8, 2024
Implemented by
Pacific Consolidated Industries

Agriculture
January 11, 2024
Implemented by
BORDA

Agriculture
January 16, 2024
Implemented by
George Greene III, PE, PhD

Agriculture
December 18, 2023
Implemented by
Clivus Multrum

Agriculture
March 7, 2024

Agriculture
December 10, 2024
Implemented by
Box Synergy

Agriculture
January 10, 2024
Implemented by
Niwa

Agriculture
January 25, 2024
Implemented by
RoadPower Inc
Have thoughts on how we can improve?
Give Us Feedback