Suggested Articles
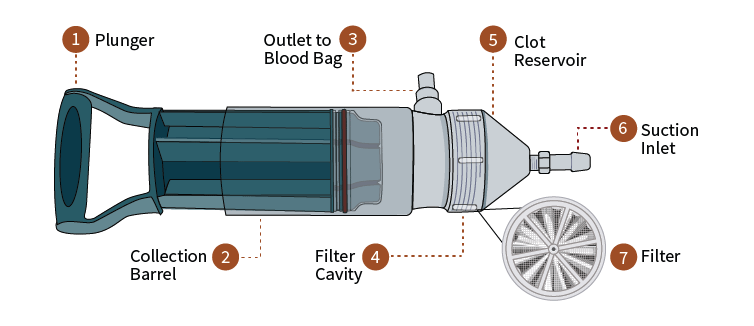
The Hemafuse blood autotransfusion device heads to clinical trials
Featured
A soup ladle is sometimes the best tool available to recycle a patient’s blood in an emergency or in a poorly equipped hospital in Sub-Saharan Africa. Health care workers can...
