
Agriculture
June 23, 2024
AguaPallet
Read SolutionImplemented by
LoooP Creative Ltd
Updated on September 26, 2024
·Created on July 31, 2019
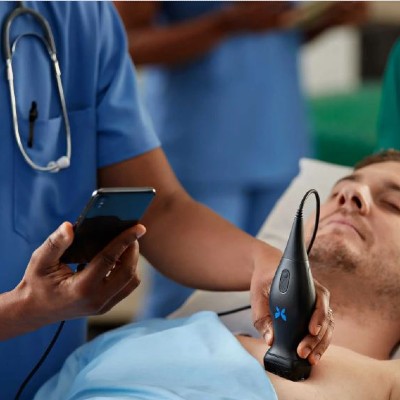
Butterfly iQ is a portable whole body ultrasound device for health diagnostics such as fetal, lung and cardiac monitoring.
The Butterfly iQ is a pocket-sized whole-body ultrasound device capable of real-time imaging with an integrated interface. Ultrasound on-chip TM technology enables the device to be portable and comes with wireless charging with backup for up to 2 hours of the continuous scan. The product is distributed under the name of Butterfly iQ. The device allows real-time collaboration with health clinics with integral storage options.
Target Countries
Australia, Canada, New Zealand, United States
Target SDGs
SDG 3: Good Health and Well-Being
Market Suggested Retail Price
$1,999.00
Target Users (Target Impact Group)
Community, Small and Medium-sized Enterprises
Distributors / Implementing Organizations
This product is available through their Reserve my iQ initiative to licensed practitioners.
Competitive Landscape
Direct competitors include Clarius Ultrasound, Healcerion Sonon 300C, and GE VScan Pocket Ultrasound.
Countries
Australia, Canada, New Zealand, United States
Manufacturing/Building Method
The product is produced in the US and made available through their website.
Intellectural Property Type
Select Type
User Provision Model
Users can obtain the product from the company's website by requesting their Reserve my iQ.
Distributions to Date Status
Since introduction, more than 30,000 Butterfly iQ and iQ+ devices have shipped to medical professionals globally.
Power Supply Type
Wireless charging 2400 mAh Lithium Ion Battery, up to 2 hours of continuous scanning
Consumables
Ultrasound gel
Indispensable equipment for function (Y/N)
Y
Maintenance or calibration required by user at time of use?
Probe cleaning and disinfection
Display Type
Digital, smartphone screen
Gestation Required for Heart Rate Detection (weeks)
First trimester of gestational age
Material of Construction
Plastic for the enclosure, Silicium and Copper for the chip and other electronics
Design Specifications
Butterfly's iQ is powered by Ultrasound-on-Chip technology, which replaces the traditional system transducer with a single silicon chip. It is portable and light, and its battery lasts approximately 2 hours of continuous work. Its main attribute is its versatiliy, having a multimode capture that enables the utilization of this device to obtain images from different human tissues. It is compatible with many mobile devices, as indicated in the user manual. Butterfly iQ is affordable in comparison to other ultrasound equipments in the market- which could cost around 200, 000 USD-, still maintaining the high quality imaging needed to an accurate diagnosis.
Hardware:
- Dimensions: 144 x 53 x 26 mm (5.6 x 2.0 x 1.0 in.)
- Weight: 313 grams (0.69 lbs)
- Cable length: 1.25 meters (4.10 feet)
- Battery: 2400 mAh Lithium Ion Battery
- Battery life: 120 minutes (2 continuous hours)
- Recharge time: 5 hours (for full recharge)
- Wireless charging: Qi-compliant
Imaging:
- Modes: M-mode, B-mode, Color Doppler
- Min/max scan depth: 2cm / 30cm
- Tools: Midline marker, Controls, Gain, TGC (near, mid, far), Depth
Presets:
Abdomen, Abdomen Deep, Aorta & Gallbladder, Bladder, Cardiac, Cardiac Deep, Fast, Lung, Musculoskeletal, Nerve, Obstetric, Pediatric Abdomen, Pediatric Cardiac, Pediatric Lung, Small Organ, Soft Tissue, and Vascular: Access, Carotid, Deep Vein.
Technical Support
Provided by the company
Replacement Components
The product does not allow replaceable components. However, the concerned user can contact the supplier for cleaning instruction and reimbursement.
Lifecycle
The product comes with a one year warranty and the life of the product depends on the number of hours of usage.
Manufacturer Specified Performance Parameters
Butterfly's Ultrasound-on-Chip technology replaces the traditional transducer and system with a single silicon chip, allowing for full-body ultrasounds with faster frame rates, optimized beamforming, and higher efficiency.
Vetted Performance Status
Butterfly iQ has CE Mark and FDA clearance for diagnostic imaging for 13 clinical indications, covered by 19 clinical presets which span the whole body: Abdominal, Cardiac, Fetal/Obstetric, Musculoskeletal (Conventional) and more. Additionally, the device demonstrate to be an effective, though imperfect, point-of-care ultrasound device in a low-resource emergency setting, after five physician used it in a emergency department over 10 weeks. The device advantages include its use of a single probe for multiple functions, small size, ease of transport, relatively low cost, and good image quality in most functions. Butterfly iQ also showed a similar performance in comparison to a traditional ultrasound after screening 53 people to find signals of shock and hypotension.
Safety
Proper cleaning and ultrasound gel should be used as per the instructions provided by the manufacturer.
Complementary Technical Systems
A reliable mobile phone or smart device with a screen is required to use the product.
Academic Research and References
Bennett, D., De Vita, E., Sestini, P., 2021, Portable Pocket-Sized Ultrasound Scanner for the Evaluation of Lung Involvement in Coronavirus Disease 2019 Patients, Ultrasound in medicine & biology, Vol. 47(1), pp. 19–24.
Convissar, D., Bittner, E. A., & Chang, M. G., 2021, Biplane Imaging Using Portable Ultrasound Devices for Vascular Access, Cureus, Vol. 13(1), pp. 12561.
Burleson, S. L., Swanson, J. F., Greene, C. J., 2020, Evaluation of a novel handheld point-of-care ultrasound device in an African emergency department, The ultrasound journal, 12(1), pp. 53.
Pafitanis G., Pawa A., 2020, The Butterly iQ: An ultra-simplified color Doppler ultrasound for bedside pre-operative perforator mapping in DIEP flap breast reconstruction, Journal of Plastic, Reconstructive & Aesthetic Surgery, Vol. 73(5), pp. 983-1007.
Sudol, W., 2010, U.S. Patent No. 7,741,756, U.S. Patent and Trademark Office.
Glüer, C. C., Eastell, R., Clowes, J., 2004, Association of five quantitative ultrasound devices and bone densitometry with osteoporotic vertebral fractures in a population?based sample: the OPUS Study, Journal of bone and mineral research, Vol. 19(5), pp. 782-793.
Pillen, S., van Dijk, J. P., Zwarts, M. J., 2009, Quantitative gray?scale analysis in skeletal muscle ultrasound: a comparison study of two ultrasound devices, Muscle & Nerve: Official Journal of the American Association of Electrodiagnostic Medicine, Vol. 39(6), pp. 781-786.
Further scientific evidence for Butterfly iQ can be found here.
Compliance with regulations
The product is CE approved and FDA cleared
Other Information
None

Agriculture
June 23, 2024
Implemented by
LoooP Creative Ltd

Agriculture
January 10, 2024
Implemented by
NRSRelief

Agriculture
December 31, 2023
Implemented by
Potential Energy

Agriculture
June 22, 2024
Implemented by
World Bicycle Relief

Agriculture
June 8, 2024
Implemented by
ClickMedix
Agriculture
February 14, 2024
Implemented by
USTAR Biotechnologies (Hangzhou) LTD

Agriculture
January 8, 2024
Implemented by
Gadgil Lab, University of California Berkeley

Agriculture
December 3, 2024
Implemented by
Envirofit International
Agriculture
February 5, 2024
Implemented by
EyeJusters

Agriculture
February 8, 2024
Implemented by
GE Healthcare
Have thoughts on how we can improve?
Give Us Feedback