
Agriculture
December 2, 2024
EcoZoom Jet
Read SolutionImplemented by
BioLite
 data coming soon
data coming soon
Updated on December 19, 2023
·Created on August 27, 2015
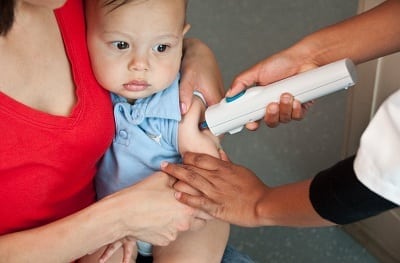
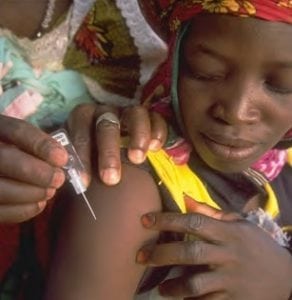
DSJI ensures primary immunization from infections using needle-free injections for developing countries.
The PATH disposable-syringe jet injector (DSJI) is a sterile needle-free device that uses a single-dose syringe and a pressurized liquid stream rather than a needle to penetrate through the skin and deliver injections to the intradermal, subcutaneous, or intramuscular tissues. The PATH DSJI aims to reduce needle reuse and the associated disease spread due to shortages of sterile needles and trained lab workers. An estimated 23.5 million new HIV, hepatitis B, and hepatitis C infections occur every year through needle reuse and accidental needle stick injuries.

Agriculture
December 2, 2024
Implemented by
BioLite

Agriculture
September 27, 2024
Implemented by
Birthing Kit Foundation
Agriculture
December 19, 2023
Implemented by
Hadleigh Health Technologies and Rice University
Agriculture
December 7, 2023
Implemented by
PATH

Agriculture
September 27, 2024
Implemented by
PATH

Agriculture
June 22, 2024
Implemented by
Phillips South Africa and Rhiza Foundation

Agriculture
December 3, 2024
Implemented by
InStove Manufacturing Ltd.

Agriculture
June 8, 2024
Implemented by
Tula Foundation

Agriculture
September 27, 2024
Implemented by
MamaOpe

Agriculture
December 4, 2023
Implemented by
Cure Tunisia
Have thoughts on how we can improve?
Give Us Feedback