Agriculture
September 26, 2024
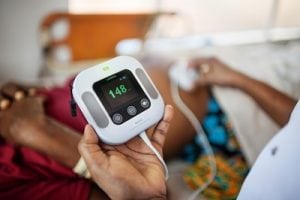
Moyo Fetal Heart Rate Monitor
Read SolutionImplemented by
Laerdal Global Health
Updated on February 5, 2024
·Created on May 25, 2016
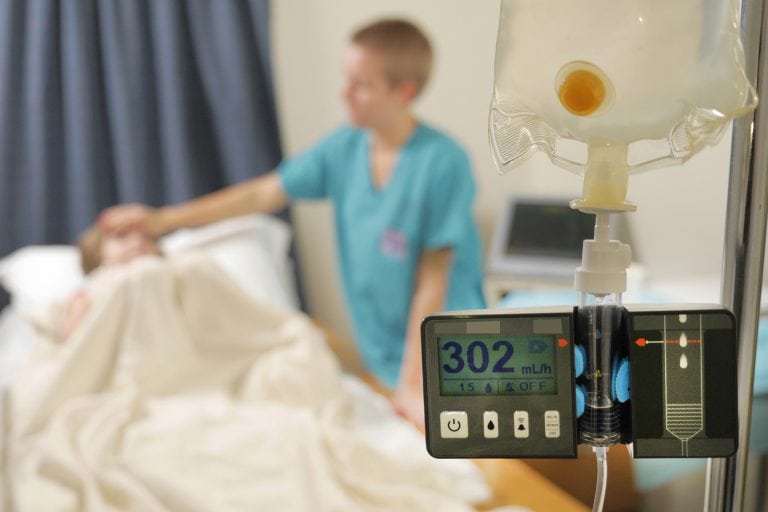
DripAssist is a rate monitor for IV delivery.
DripAssist is an infusion rate monitor that clips onto any gravity drip set to continuously monitor flow rate and volume when administering IV medication.
Information for this product was provided courtesy of the WHO’s 2016 Call for Innovative Health Technologies for Low-Resource Settings.
Target SDGs
SDG 3: Good Health and Well-Being
Market Suggested Retail Price
$395.00
Target Users (Target Impact Group)
Small and Medium-sized Enterprises, Public Sector Agencies
Distributors / Implementing Organizations
Shift Labs. Further distributtors coming soon. Interview with representative
Competitive Landscape
Direct competitors include Rice University IV DRIP.
Regions
Worldwide
Manufacturing/Building Method
Manufactured in USA under specific conditions required to maintain regulatory compliance of class II medical device according to FDA classifications (11b for CE).
Intellectural Property Type
Patent
User Provision Model
Users can obtain the product/service from the manufacturer, national distributor, international distributor, and direct sales.
Distributions to Date Status
1,000-10,000 units Interview with representative
Maximum dose volume monitored
999mLInterview with representative
Measurement units
Flow rate (mL/h), drops per minute (dp/m) and total volume (mL)
Accuracy
±1% drip rate
Alarm (Y/N)
Yes, 89 dB at 10cm approx. Sounds at ±13% rate change or when flow stops.
Consumables
None
Power supply type
One AA battery
Indispensable equipment for function (Y/N)
Yes, I.V drip and medication
Maintenance or calibration required by user at time of use? (Y/N)
No
Technical Support
Provided by in country distributor or call/email info@shiftlabs.com.
Webinars are available for DripAssist training. Interview with representative
Replacement Components
One AA battery, which the user should be able to find in his or her country of residence ^(Interview with representative)
Lifecycle
2+ years Interview with representative
Battery life on a full charge is 360-400 hours. Interview with representative.
Manufacturer Specified Performance Parameters
To solve a problem that nurses and doctors reported having-- administering medication accurately with gravity. ^(Interview with representative)
Vetted Performance Status
Nurses and doctors at international health organizations and hospitals report being able to administer medication accurately with DripAssist Interview with representative.
Safety
Certain safety precautions can be found in a user manual
Complementary Technical Systems
I.V. drip, medication
Academic Research and References
Raghavendra B ; Vijayalakshmi K ; Manish Arora, Intravenous Drip Meter and Controller, IEEE Xplore: 24 March 2016
Shift Labs. Device, Method and System for Monitoring the delivery of fluids through a drip chamber, Patent, 2015.
Shift Labs, “DripAssist Webinar – Learn to Use DripAssist in One Minute“, Youtube, 2017
Compliance with regulations
FDA-cleared and CE-marked
IEC 60601-1: 2005 + CORR. 1:2006 + CORR. 2:2007 + AM1:2012
ANSI/AAMI ES60601-1:2005+A2 (R2012)
Evaluation methods
Field tested in Rwanda, West Africa, and Haiti. Interview with representative.
Other Information
Unknown.

Agriculture
September 26, 2024
Implemented by
Laerdal Global Health
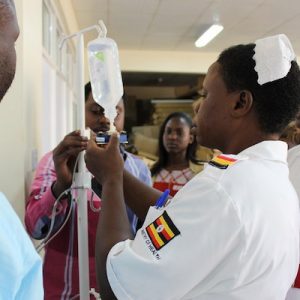
Agriculture
December 4, 2023
Implemented by
Uganda Industrial Research Institute
Agriculture
March 11, 2024
Implemented by
Capnia
Agriculture
September 27, 2024
Implemented by
Neopenda

Agriculture
March 9, 2024
Implemented by
Laerdal Global Health

Agriculture
January 8, 2024
Implemented by
DayOne Response
Agriculture
December 19, 2023
Implemented by
Hadleigh Health Technologies and Rice University

Agriculture
November 30, 2024
Implemented by
El Fuego del Sol
Agriculture
February 5, 2024
Implemented by
GE Healthcare

Agriculture
June 22, 2024
Implemented by
Hippo Roller
Have thoughts on how we can improve?
Give Us Feedback