Agriculture
September 26, 2024
Moyo Fetal Heart Rate Monitor
Read SolutionImplemented by
Laerdal Global Health

Updated on March 9, 2024
·Created on August 30, 2021
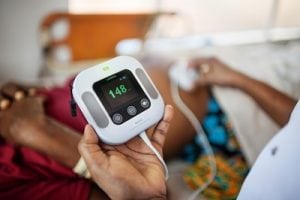
A reusable neonatal ECG heart rate meter. Courtesy of WHO Compendium 2021
NeoBeat is a reusable, consumable-free heart rate meter that provides an accurate and continuous display of newborn heart rate. The device takes seconds to put on a newborn’s abdomen, and the instantaneous heart rate can help guide neonatal resuscitation.
This product was selected for inclusion in WHO’s 2021 Compendium of Innovative Health Technologies for Low‐Resource Settings.
Target SDGs
SDG 3: Good Health and Well-Being
Market Suggested Retail Price
$149.99
Target Users (Target Impact Group)
Household, Small and Medium-sized Enterprises, NGOs
Distributors / Implementing Organizations
Laerdal Global Health
Competitive Landscape
Direct competitors include Spandan ECG.
Regions
Africa
Manufacturing/Building Method
NeoBeat is manufactured in Norway.
Intellectural Property Type
Patent
User Provision Model
The product can be purchased directly from the manufacturer.
Distributions to Date Status
Between 101-1000 distributions as of October 2020.
Alert mechanism
No
Clinical measurements monitored
Heartrate
Patient population
Newborns
Maximum duration of consecutive usage
Unknown
Design Specifications
The device uses dry stainless steel electrodes (recommended by ILCOR 2015 guidelines) embedded in a spring-elastic buckle to read ECG signals from newborns. To use the device, remove it from the charging stand, spread the electrode arms slightly, and apply over the newborn's chest. After ~5 seconds, the device will display the newborn's ECG heart rate. The product includes a rechargeable battery, signals processor, and LED-display, and weighs 0.031 kg.
Technical Support
Provided by the manufacturer.
Replacement Components
Replacement components are available separately through the manufacturer.
Lifecycle
The device has a lifetime of 2-5 years, and a 1-year warranty is provided with purchase. The battery has a lifetime of 3-6 years.
Manufacturer Specified Performance Parameters
The manufacturer specifies that the device is easy to use and maintain, accurate, affordable, and durable.
Vetted Performance Status
Neonate was tested for usability in Norway, India, Tanzania, and the UK with gynecologists, pediatricians, midwives, and pediatric nurses. Testing by University Hospital in Norway found that the device conforms with IEC 62366 and CE-marking.
Safety
This device should only be used by trained professionals. Additionally, the device should be disinfected with 70% ethanol between uses.
Complementary Technical Systems
Electricity to recharge batteries
Academic Research and References
Bush J.B., Cooley V., Perlman J., Chang C., 2021, NeoBeat offers rapid newborn heart rate assessment. Arch Dis Child Fetal Neonatal Ed. doi: 10.1136/archdischild-2020-320901. Epub ahead of print. PMID: 33478958
Lee A., et al., 2013, Intrapartum-related neonatal encephalopathy incidence and impairment at regional and global levels for 2010 with trends from 1990. Pediatric Research, Vol. 74(s1).
Linde J., et al., 2016, Normal Newborn Heart Rate in the First Five Munites of Life Assessed by Dry-Electrode Electrocardiography. Neonatology, Vol. 110(3), pp. 231-237.
Compliance with regulations
This device complies with the following standards: IEC 60601-1, IEC 60601-1-6, IEC 62304, IEC 62366-1, and ISO 10993, and is CE-marked.
Evaluation methods
This product has been evaluated for usability in studies in Nepal, Tanzania, Uganda, and DR Congo and is part of the Safer Births research program.
Other Information
None

Agriculture
September 26, 2024
Implemented by
Laerdal Global Health

Agriculture
November 22, 2023
Implemented by
Laerdal Global Health

Agriculture
September 27, 2024
Implemented by
Design That Matters (DtM)

Agriculture
January 17, 2024
Implemented by
Extech Instruments

Agriculture
February 5, 2024
Implemented by
Shift Labs

Agriculture
February 6, 2024
Implemented by
Toray Industries Inc.

Agriculture
June 22, 2024
Implemented by
Wello

Agriculture
January 3, 2024
Implemented by
Suprio Das

Agriculture
January 2, 2024
Implemented by
Archimedes Project

Agriculture
November 22, 2024
Implemented by
Green Bio Energy
Have thoughts on how we can improve?
Give Us Feedback