
Agriculture
November 30, 2024
AGNI Star Rice Husk Gas Stove
Read SolutionImplemented by
NDMI Renewable Energy Private Limited
Updated on February 5, 2024
·Created on August 1, 2019
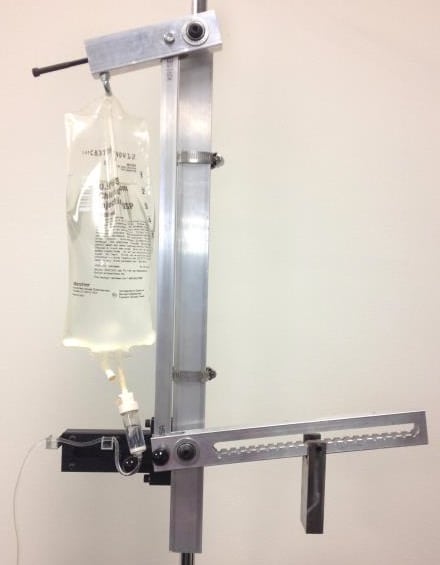
The IV DRIP is a monitor created by engineers at Rice University to monitor intravenous dehydration relief.
The IV DRIP is a monitor for intravenous dehydration relief in pediatric medicine. It is a completely mechanical device to monitor the volume flow rate from the IV. This product is in prototype phase.
Target SDGs
SDG 3: Good Health and Well-Being
Market Suggested Retail Price
$80.00
Target Users (Target Impact Group)
Small and Medium-sized Enterprises, Public Sector Agencies
Distributors / Implementing Organizations
Unknown
Competitive Landscape
Direct competitors include DripAssist Infusion Rate Monitor.
Regions
Africa
Manufacturing/Building Method
IV drip is currently in the prototype stage. It was constructed by students at Rice University out of steel, aluminum, and plastic components with machinery found in a standard machine shop.
Intellectural Property Type
Select Type
User Provision Model
The designers have not yet selected their user provision model.
Distributions to Date Status
None
Maximum dose volume monitored
800 mL
Measurement units
mL
Accuracy
97.5%
Alarm (Y/N)
No
Consumables
None
Power supply type
None
Indispensable equipment for function (Y/N)
None
Maintenance or calibration required by user at time of use? (Y/N)
Yes
Design Specifications
The IV DRIP is a completely mechanical device for monitoring flow by regulating volume discharge. The flow is controlled using a system of counterweights which is based upon the design of a mousetrap. This system creates a kink in the IV tubing in order to stop fluid flow. It also ensures that the IV bag remains sterile.
Technical Support
None available. This product is a prototype.
Replacement Components
None
Lifecycle
Approximately 10 years if used twice weekly
Manufacturer Specified Performance Parameters
Vetted Performance Status
The device is in the testing stage at hospitals in Malawi. Clinicians reviewed the product as "easy to use". The designers also did durability testing on the device, simulating 1000 uses, and found that the device stood up to the test.
Safety
The devices is 97.5% accurate, in sever cases this may not be accurate enough.
Complementary Technical Systems
IVs
Academic Research and References
There is no academic research at this time. The full design report can be found here.
Compliance with regulations
Unknown
Evaluation methods
User interviews and trials at hospitals in Malawi.
Other Information
None

Agriculture
November 30, 2024
Implemented by
NDMI Renewable Energy Private Limited

Agriculture
September 18, 2024
Implemented by
Project Support Services (PSS)

Agriculture
August 21, 2024
Implemented by
EOS International

Agriculture
January 22, 2024
Implemented by
Proximity Designs

Agriculture
January 20, 2024
Implemented by
SunCulture

Agriculture
August 20, 2024
Implemented by
Jain Irrigation Systems Ltd.

Agriculture
August 21, 2024
Implemented by
Illuminum Greenhouses Kenya

Agriculture
December 7, 2023
Implemented by
Katadyn Group

Agriculture
February 22, 2024
Implemented by
Greenhouse Ventures

Agriculture
February 20, 2024
Implemented by
Stone Cold Systems
Have thoughts on how we can improve?
Give Us Feedback