
Agriculture
June 23, 2024
AguaPallet
Read SolutionImplemented by
LoooP Creative Ltd

Updated on February 15, 2024
·Created on September 26, 2019
PointCheck is a portable, fast, and non-invasive white cell monitoring device.
The device works on the principle of imaging the blood flow and counting the number of white blood cells. PointCheck™ is a non-invasive device which allows immunosuppressed patients to get their blood check done. The device will potentially help health clinics in having more frequent and rapid testing done. Currently this is an investigational device and it is not for sale.
Target SDGs
SDG 3: Good Health and Well-Being
Target Users (Target Impact Group)
Small and Medium-sized Enterprises, Public Sector Agencies
Distributors / Implementing Organizations
The company is supported by MIT Innovation Initiative, the MIT Deshpande Center for Technological Innovation, the MIT Sandbox Innovation Fund, the Martin Trust Center for Entrepreneurship, the MIT Translational Fellows Program, and the MIT Venture Mentoring Service.
Countries
Spain, United States
Manufacturing/Building Method
Leuko is working on developing prototypes and commercializing the technology with help from the MIT Innovation Initiative, the MIT Deshpande Center for Technological Innovation, the MIT Sandbox Innovation Fund, the Martin Trust Center for Entrepreneurship, the MIT Translational Fellows Program, and the MIT Venture Mentoring Service.
Intellectural Property Type
Patent
User Provision Model
This product is in the prototype phase and is not available for distribution yet.
Distributions to Date Status
None
Consumables
Unknown
Detection sensitivity
Unknown
Indispensable equipment for function (Y/N)
Unknown
Maintenance or calibration required by user at time of use? (Y/N)
Yes
Number of Tests Performed
Unknown
Power supply type: Continuous, Recharging only (V, time required, battery life), Other
Unknown
Time required for procedure (minutes)
Unknown
Design Specifications
The design of the device allows chemotherapy patients to prevent infections from needle-based tests. The device uses imaging technique which is non-invasive and thus quick check on white blood cell count can be done. The device is portable enough to be used in a clinic or home applications where the patient can have access to daily checks on their blood count. The device is enclosed in a plastic casing and is suitable for use in the home, clinic, or healthcare centers.
PointCheck™ is able to determine if white cell levels are dangerously low by imaging the blood flowing through the capillaries in the finger. PointCheck does this without having to extract any blood
Technical Support
Not yet available as product is in prototype stage
Replacement Components
The replaceable component includes the battery
Lifecycle
2-5 years
Manufacturer Specified Performance Parameters
Accurately measure white blood cell levels.
Vetted Performance Status
The team showed that the device could accurately determine whether white blood cell levels were too low, in a study of 11 patients undergoing chemotherapy.
Safety
No known safety hazards are related to this product.
Complementary Technical Systems
A reliable power source is required for the device to function and read the blood flow.
Academic Research and References
Tajiri, J., Noguchi, S., Murakami, T. and Murakami, N., 1990, Antithyroid drug–induced agranulocytosis: the usefulness of routine white blood cell count monitoring, Archives of internal medicine, 150(3), pp. 621-624.
Bates, M.F., Khander, A., Steigman, S.A., Tracy, T.F. and Luks, F.I., 2014, Use of white blood cell count and negative appendectomy rate, Pediatrics, 133(1), pp. e39-e44.
Van’t Oever, R., et al, 2003, “Optical method and apparatus for red blood cell differentiation on a cell-by-cell basis, and simultaneous analysis of white blood cell differentiation.“, U.S. Patent No. 6,630,990.
Pablo-Trinidad, A., Butterworth, I., Ledesma-Carbayo, M.J., Vettenburg, T., Sánchez-Ferro, Á., Soenksen, L., Durr, N.J., Barrutia, A.M., Cerrato, C., Humala, K. and Urdiol, M.F., 2019. “Automated detection of neutropenia using noninvasive video microscopy of superficial capillaries.“ American journal of hematology, 94(8), p.E219.
Bourquard, A., Pablo-Trinidad, A., Butterworth, I., Sánchez-Ferro, Á., Cerrato, C., Humala, K., Urdiola, M.F., Del Rio, C., Valles, B., Tucker-Schwartz, J.M. and Lee, E.S., 2018. “Non-invasive detection of severe neutropenia in chemotherapy patients by optical imaging of nailfold microcirculation.“ Scientific reports, 8(1), pp.1-12.
Trafton, Anne. “Monitor detects dangerously low white blood cell levels“, MIT News, 2018
Compliance with regulations
None yet as product is in prototype stage
Evaluation methods
The product was tested for its accuracy in detecting low white blood cell levels.
Other Information
None

Agriculture
June 23, 2024
Implemented by
LoooP Creative Ltd

Agriculture
January 10, 2024
Implemented by
NRSRelief

Agriculture
December 31, 2023
Implemented by
Potential Energy

Agriculture
June 22, 2024
Implemented by
World Bicycle Relief

Agriculture
June 8, 2024
Implemented by
ClickMedix
Agriculture
February 14, 2024
Implemented by
USTAR Biotechnologies (Hangzhou) LTD

Agriculture
January 8, 2024
Implemented by
Gadgil Lab, University of California Berkeley

Agriculture
December 3, 2024
Implemented by
Envirofit International
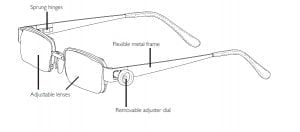
Agriculture
February 5, 2024
Implemented by
EyeJusters

Agriculture
February 8, 2024
Implemented by
GE Healthcare
Have thoughts on how we can improve?
Give Us Feedback