
Agriculture
November 29, 2023
AT-Knee
Read SolutionImplemented by
LegWorks

Updated on December 1, 2023
·Created on August 27, 2015
Artificial knee joint designed for above-knee amputees in the developing world
The D-Rev ReMotion Knee is an artificial knee joint designed for above-knee amputees in the developing world. The product was selected for inclusion in WHO’s 2013 Compendium of Innovative Health Technologies for Low-Resource Settings.
Target SDGs
SDG 3: Good Health and Well-Being
Market Suggested Retail Price
$80.00
Target Users (Target Impact Group)
Household, Community
Distributors / Implementing Organizations
D-Rev and partner prosthetics providers, specifically BMVSS in India.
Competitive Landscape
Direct competitors include AT-Knee and Jaipur Foot.
Countries
Ecuador, Guatemala, India, Indonesia, Iraq, Liberia, Pakistan, Sudan, Tanzania, Uganda
Manufacturing/Building Method
The ReMotion Knee is mass produced by a contract manufacturer in China.Interview with representative
Intellectural Property Type
Patent
User Provision Model
D-Rev partners with in-country prosthetic providers with already established distribution channels and local expertise. Users will be able to purchase the ReMotion Knee from these providers.
Distributions to Date Status
The ReMotion Knee became commercially available in December 2015. Field trials included deployment in three countries with 103 amputees wearing the v3.
As of April 2015, 6,800 patients have been fitted with the v3 predecessor JaipurKnee by BMVSS.
Craftsmanship required
Skilled prosthetisist
Patient satisfaction
86%
Patient compliance
Unknown
Comfort
Unknown
Materials
Acetal (polyoxymethylene) and stainless steel
Water resistant (yes/no)
Yes
Amputation level
Transfemoral
Weight (kg)
0.618 kg
Design Specifications
Specifications for the ReMotion Knee v3 are - Water resistant: withstands humid and wet climates, without rusting or swelling - Materials of construction: High strength polymers and stainless steel components. - Weight limit of 80 kg (176 pounds) - Knee Weight: 618 g (22 oz) - Range of motion: 160 degrees, enabling kneeling, squatting, and biking - Noise Dampening: reduces noise during walking - Works with standard prosthetic leg systems
Technical Support
A trained clinician is required to custom mold the socket to fit the patient and assemble it with the other components. D-Rev partners with clinics that are staffed with prosthetics experts to provide support to patients.
Replacement Components
Available from D-Rev
Lifecycle
ReMotion Knee v3 has a lifetime of 3-5 years.
Manufacturer Specified Performance Parameters
D-Rev performance measures for the ReMotion Knee v3 include: - stable gait with a natural swinging motion, high range of motion and effect noise dampening - percentage of amputees fit who are still wearing their knee six months after the fitting (“Compliance Rate”) - percentage of amputees who report experiencing no mechanical failures of their knee (“Non-failure Rate”) - percentage of amputees fit who report being “satisfied” or “very satisfied” with their knee (“Satisfaction Rate”)
Targets have not been indicated.
Vetted Performance Status
ReMotion officially passed all ISO 10328 testing standard for lower-limb prosthetics and has received CE mark.
Safety
The ReMotion Knee must be fitted by a trained clinician to ensure patient safety.
Complementary Technical Systems
Standard prosthetic leg systems containing a foot, a pylon, and a socket. The knee joint is typically the most expensive and complex component of a full prosthesis.
Academic Research and References
Compliance with regulations
The ReMotion Knee meets ISO 10328 standards of wear simulation and has received CE mark. CE mark certificate is available upon request.
Evaluation methods
Field trials with prosthetic clinics. The Jakarta School of Prosthetics and Orthotics (JSPO) and PUSPADI Bali (formerly Yakkum Bali) in Indonesia fit the first patients with the knee in May 2013 and D-Rev launched a 6-month follow-up study. D-Rev aimed to learn about the effectiveness of the v3 design, what features need tweaking before full-scale production, and how the knee impacts the lives of the patients. D-Rev also collected feedback from the prosthetists fitting the knee whose opinions and adoption of the knee are just as critical for success as those of the patients.Structural analysis of the
ReMotion was achieved via finite element analysis (FEA) and 1-meter drop test.

Agriculture
November 29, 2023
Implemented by
LegWorks

Agriculture
June 23, 2024
Implemented by
LoooP Creative Ltd

Agriculture
January 10, 2024
Implemented by
NRSRelief

Agriculture
December 31, 2023
Implemented by
Potential Energy

Agriculture
June 22, 2024
Implemented by
World Bicycle Relief

Agriculture
June 8, 2024
Implemented by
ClickMedix
Agriculture
February 14, 2024
Implemented by
USTAR Biotechnologies (Hangzhou) LTD

Agriculture
January 8, 2024
Implemented by
Gadgil Lab, University of California Berkeley

Agriculture
December 3, 2024
Implemented by
Envirofit International
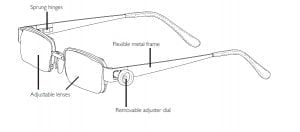
Agriculture
February 5, 2024
Implemented by
EyeJusters
Have thoughts on how we can improve?
Give Us Feedback