
Agriculture
November 22, 2023
Laerdal Upright Newborn Bag-Mask
Read SolutionImplemented by
Laerdal Global Health
Updated on March 8, 2024
·Created on August 31, 2021
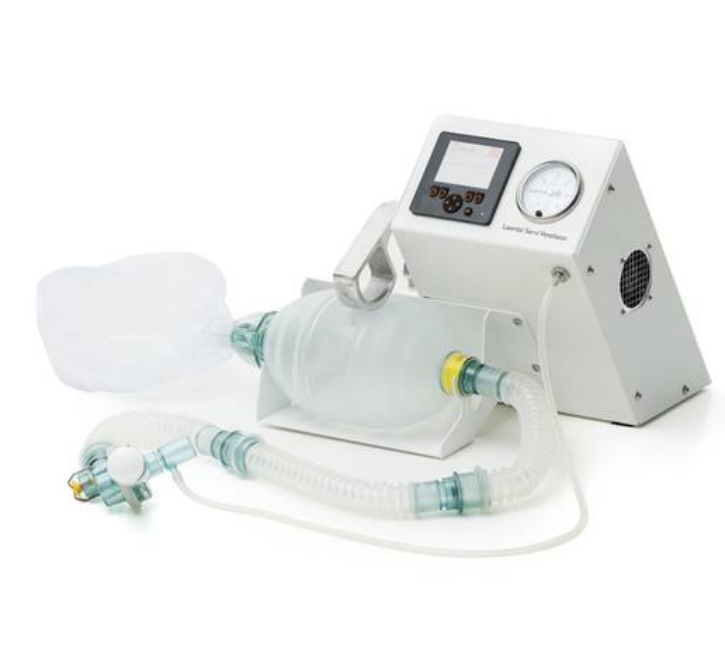
The Laerdal Servi Ventilator is an emergency invasive ventilator designed and implemented in response to the COVID-19 pandemic. Courtesy of WHO Compen
Developed in 2020 in response to the needs from the global COVID-19 pandemic, the Laerdal Servi Ventilator is an emergency invasive ventilator designed not to replace existing ventilators in over-capacity hospitals, but to offer a supplemental technology in emergency situations. As such, the design features settings for sedated patients with respiratory failure, intended to be used over a relatively short period of time on the patient.
This product was selected for inclusion in WHO’s 2021 Compendium of Innovative Health Technologies for Low‐Resource Settings.
Target SDGs
SDG 3: Good Health and Well-Being
Target Users (Target Impact Group)
Small and Medium-sized Enterprises, Public Sector Agencies
Distributors / Implementing Organizations
This product was implemented by the following organizations: the Norwegian Defense Research Establishment, Edge Health Technologies AS, Laerdal Medical AS, and Servi AS.
Competitive Landscape
Direct competitors include Gradian Comprehensive Care Ventilator (CCV).
Regions
Europe
Countries
Norway, United Kingdom
Manufacturing/Building Method
The product is manufactured by Laerdal in Stavanger, Norway. The company produces components and and other products from their factories in Norway, Mexico, China, and USA.
Intellectural Property Type
Select Type
User Provision Model
The emergency ventilators are purchased by European governments to then distribute to hospitals in need.
Distributions to Date Status
1000 ventilators as of 2020
Use case (category and target population)
Intensive Care; adult (? 40 kg)
Ventilation modes
VCV
Internal PEEP capability and range
No (External Disposable PEEP Valve)
Range pressure setting
Not applicable
Peak pressure limitation
Yes
FiO2 settings
Fixed, External, Not Measured
Inspiratory/expiratory pause maneuver
No
Primary display modes
PEEP, tidal volume, ventilator mode status
Spirometry available
No
Capnography available
No
Main alarms available
No (High/low Mv and High/low FiO not present)
Air source, O2 low pressure capability
Internal (Automated Resuscitation Bag), Yes
Internal or external battery (operating time, recharging time)
Internal (5 hours, 8 hours)
Power requirements (W)
270 W
Design Specifications
The Laerdal Servi Ventilator incorporates the following components: (1) oxygen tubing and reservoir kit, (2) three 28 cm extension tubes, (3) patient valve with pressure relief valve, (4) exhalation port with a diameter of 30 mm, and (5) manometer connector. There are three consumables: (6) HME filter, (7) Thomas Tube holder, and (8) adjustable PEEP valve. The control unit allows for tidal volume adjustment between 100-800 ml and ventilation rates between 5-30 per min.
Technical Support
Technical support is provided by the manufacturer.
Replacement Components
Replacement components and consumables are available for purchase from the manufacturer
Lifecycle
Unknown
Manufacturer Specified Performance Parameters
The manufacturer's goal of the rapid-development project emphasized quick production, simple to use, and simplified (but accurate) automated assisted breathing.
Vetted Performance Status
The system has been tested with test lungs to in both controlled and assisted ventilation mode, details of the performance testing and design iterations are available in the FFI report.
Safety
The product has been designed and tested to meet patient safety standards in Norway.
Complementary Technical Systems
None
Academic Research and References
FFI-Report, Rapid innovation run for the health sector: development of an emergency respirator in connection with the coronavirus crisis, 2020
Laerdal Global Health, “Laerdal Servi Ventilator“, 2020
Compliance with regulations
Received the European CE (Communauté Européenne) Mark
Evaluation methods
Unknown
Other Information
None

Agriculture
November 22, 2023
Implemented by
Laerdal Global Health

Agriculture
September 27, 2024
Implemented by
Diamedica

Agriculture
February 2, 2024
Implemented by
Gradian Health Systems

Agriculture
March 8, 2024
Implemented by
MTTS

Agriculture
March 8, 2024
Implemented by
Mechanical Ventilator Milano

Agriculture
March 8, 2024
Implemented by
Conseil Européen pour la Recherche Nucléaire (CERN)

Agriculture
December 29, 2023
Implemented by
Lifesaver

Agriculture
November 30, 2024
Implemented by
Sustainable Community Development Services (SCODE)

Agriculture
February 21, 2024

Agriculture
February 15, 2024
Implemented by
Leuko
Have thoughts on how we can improve?
Give Us Feedback