Agriculture
November 24, 2023
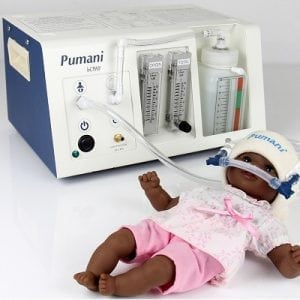
Pumani Bubble Continuous Positive Airway Pressure (bCPAP) Device
Read SolutionImplemented by
Hadleigh Health Technologies, and Rice 360

Updated on November 22, 2023
·Created on August 27, 2015
A respiratory support device for infants.
The Seattle Children’s Hospital High Oscillation Continuous Positive Airway Pressure (Sea-PAP) Device is a respiratory support technology for infants. The NeoRest Team claims that Sea-PAP improves upon conventional bCPAP by more consistently producing a range of oscillations thought to improve lung function and make it easier for an infant to breathe.
Target Regions
Africa, Central Asia, East Asia, North Asia, South America, South Asia, Southeast Asia
Target SDGs
SDG 3: Good Health and Well-Being
Target Users (Target Impact Group)
Community, Small and Medium-sized Enterprises
Distributors / Implementing Organizations
The NeoRest Team is in the process of finalizing the Sea-PAP device design, identifying manufacturers to produce the device and preparing to conduct clinical trials. It is not being distributed widely at this time.
Regions
Africa, Central Asia, East Asia, North Asia, South America, South Asia, Southeast Asia
Manufacturing/Building Method
The main Sea-PAP device body will be mass produced. The unit can be assembled on-site with a small tool kit of supplies including a continuous air supply, a breathing circuit tube, a tube for the nose and water.
Intellectural Property Type
Other
User Provision Model
Unknown
Distributions to Date Status
Prototype device which has not been distributed at scale.
Design Specifications
The Sea-PAP is intended to provide continuous positive airway pressure (CPAP) to spontaneously breathing neonates and infants, up to weights of 10 kg, requiring respiratory support due to conditions associated with prematurity, such as Respiratory Distress Syndrome, or other conditions where CPAP is required and/or prescribed by a physician.Sea-PAP works similarly to conventional Bubble CPAP (bCPAP) devices in providing a continuous flow of air via tubes inserted in an infant's nostrils. The Sea-PAP is comprised of two key elements; a water container and a tube inserted into the water. The Water Reservoir contains the water and has a lid permanently attached to help prevent splashing and spillages. The lid has an integrated Water Level Adjustment Port, to allow a clinician to adjust the water level in the Reservoir, if necessary, without disconnecting the breathing circuit. The tube is comprised of a Funnel-Swivel that is permanently attached to Bubble Tube. The Funnel-Swivel helps prevent spillages when filling the Water Reservoir initially and can rotate to help reduce tension that may build up in the expiratory limb of the breathing circuit. The expiratory end of the patient's breathing circuit, distal to the patient, is inserted into the center of the Funnel-Swivel. A Bubble Tube Lock mechanism is used to ensure the depth of the tube in the water does not change unintentionally. The gas bubbling out of the end of the Tube creates air pressure oscillations in the breathing circuit. For the Sea-PAP device the breathing circuit tube is placed in water at a 135 degree slope, which creates fluctuations in the positive airway pressure and keep the lungs open, lessening the work required from respiratory muscles and making it easier for the infant to breathe.
Technical Support
N/A
Replacement Components
Unknown
Lifecycle
The breathing and nose tubes can be used multiple times if sterilized. Disposable units can be used, however require a robust consumables supply chain.
Manufacturer Specified Performance Parameters
Unknown
Vetted Performance Status
Unknown
Safety
Unknown
Complementary Technical Systems
The Hansen Ventilator - a separate respiratory device - can be used in conjunction to treat even more premature infants with respiratory distress.
Academic Research and References
Diblasi RM, Noninvasive respiratory support of juvenile rabbits by high-amplitude bubble continuous positive airway pressure. Pediatr Res. 2010 Jun;67(6):624-9. doi: 10.1203/PDR.0b013e3181dcd580.
Diblasi RM, Nasal continuous positive airway pressure (CPAP) for the respiratory care of the newborn infant. Respir Care. 2009 Sep;54(9):1209-35.
Compliance with regulations
In October 2013, Sea-PAP received clearance from the Food and Drug Administration for use and distribution in the U.S.
Evaluation methods
The Seattle-PAP Bubble Nasal CPAP and Work of Breathing clinical trial is currently underway.
Other Information
Unknown

Agriculture
November 24, 2023
Implemented by
Hadleigh Health Technologies, and Rice 360

Agriculture
February 20, 2024
Implemented by
Intellectual Ventures Lab

Agriculture
March 10, 2024
Implemented by
Disease Diagnostic Group
Agriculture
February 8, 2024
Implemented by
Design That Matters (DtM)

Agriculture
January 3, 2024
Implemented by
Stanford Researchers and the International Centre for Diarrhoeal Disease Research, Bangladesh

Agriculture
February 5, 2024
Implemented by
Circ MedTech
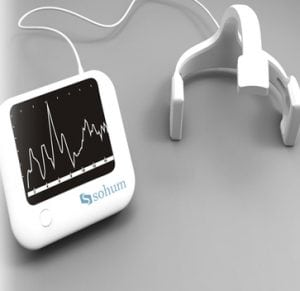
Agriculture
December 12, 2023
Implemented by
Sohum Innovation lab
Agriculture
December 19, 2023
Implemented by
Star Syringe

Agriculture
September 26, 2024
Implemented by
Jorge Odón

Agriculture
February 26, 2024
Implemented by
Hemex Health
Have thoughts on how we can improve?
Give Us Feedback